 |
|||
|
|
|||
|
|
|||
| ||||||||||
|
|  DOE-HDBK-1130-98
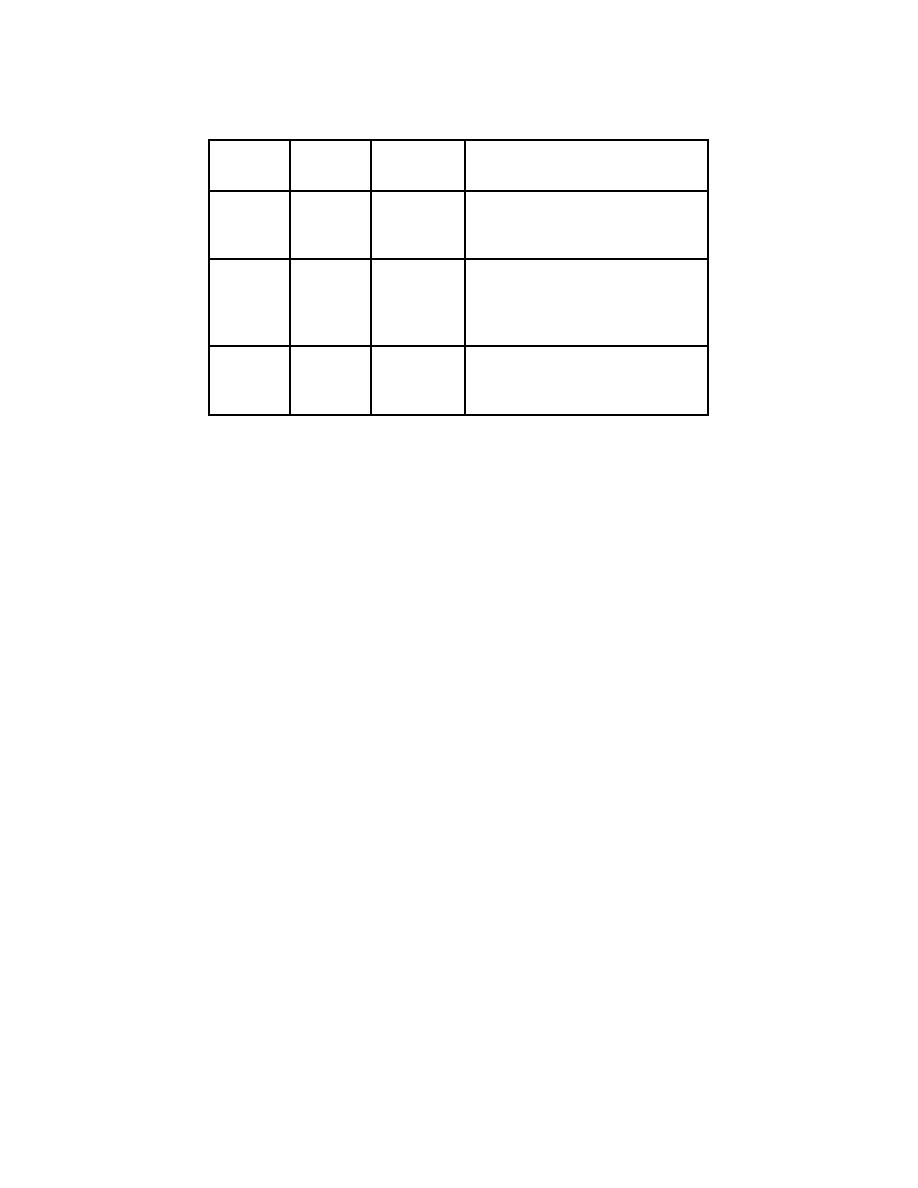
Basic Particles
3 Basic
Location
Charge
Comments
Particles
Protons
Nucleus
+ (positive)
Number of protons determines the
element. If the number of protons
changes, the element changes.
Neutrons
Nucleus
No Charge
Atoms of the same element have the
same number of protons, but can
have a different number of neutrons.
This is called an isotope.
Electrons
Orbit
- (negative)
This negative charge is equal in
nucleus
magnitude to the proton's positive
charge.
2.
Stable and unstable atoms
Only certain combinations of neutrons and protons result in stable atoms.
a.
If there are too many or too few neutrons for a given number of protons, the nucleus
will not be stable.
b.
The unstable atom will try to become stable by giving off excess energy. This energy is
in the form of particles or rays (radiation). These unstable atoms are known as
radioactive atoms.
3.
Charge of the atom
The number of electrons and protons determines the overall electrical charge of the atom.
The term "ion" is used to define atoms or groups of atoms that have a net positive or
negative electrical charge.
a.
No charge (neutral)
If the number of electrons equals the number of protons, the atom is electrically neutral.
This atom does not have a net electrical charge.
b.
Positive charge (+)
If there are more protons than electrons, the atom is positively charged.
c.
Negative charge (-)
If there are more electrons than protons, the atom is negatively charged.
4
|
|
Privacy Statement - Press Release - Copyright Information. - Contact Us |