 |
|||
|
|
|||
|
Page Title:
Physical and Chemical Properties |
|
||
| ||||||||||
|
| 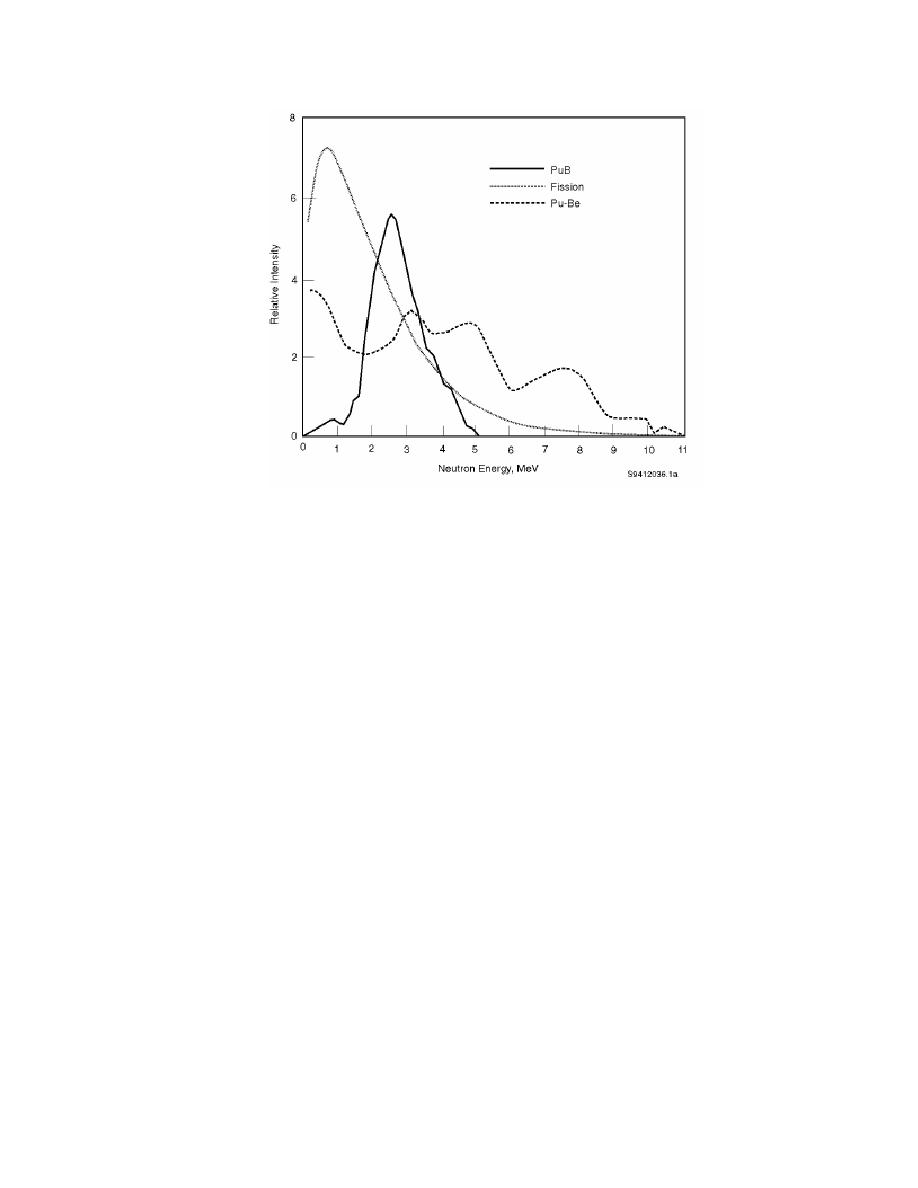 DOE-STD-1128-98
Figure 2.2.
Function of Time Since Chemical Separation
2.3
PHYSICAL AND CHEMICAL PROPERTIES
This discussion of plutonium's physical and chemical properties begins with
plutonium metal, followed by its alloys and compounds. Knowledge of the physical
properties of these classes of materials and how the plutonium was produced is the
key to understanding and predicting the hazards of working with this challenging
element. According to Healy (1993), "Nature does not decide what happens to any
material based on its radioactivity but rather on its form and mass." Form and mass
are determined by the engineering application and the kinds of processes needed to
achieve both intermediate and final products. Thus, to prevent nature from taking its
course, there can be no shortcuts in good practices for plutonium facilities.
2.3.1 Plutonium Metal
The metallic state of plutonium is undoubtedly the most complicated of all the
elements. Plutonium is a silvery-white metal, much like nickel in appearance. It
has a low melting point (640C) and an unusually high boiling point (3327C).
The metal exists in six allotropic forms, as indicated in Table 2.5. Two of the
upon heating. At room temperature, pure
2-9
|
|
Privacy Statement - Press Release - Copyright Information. - Contact Us |