 |
|||
|
|
|||
|
Page Title:
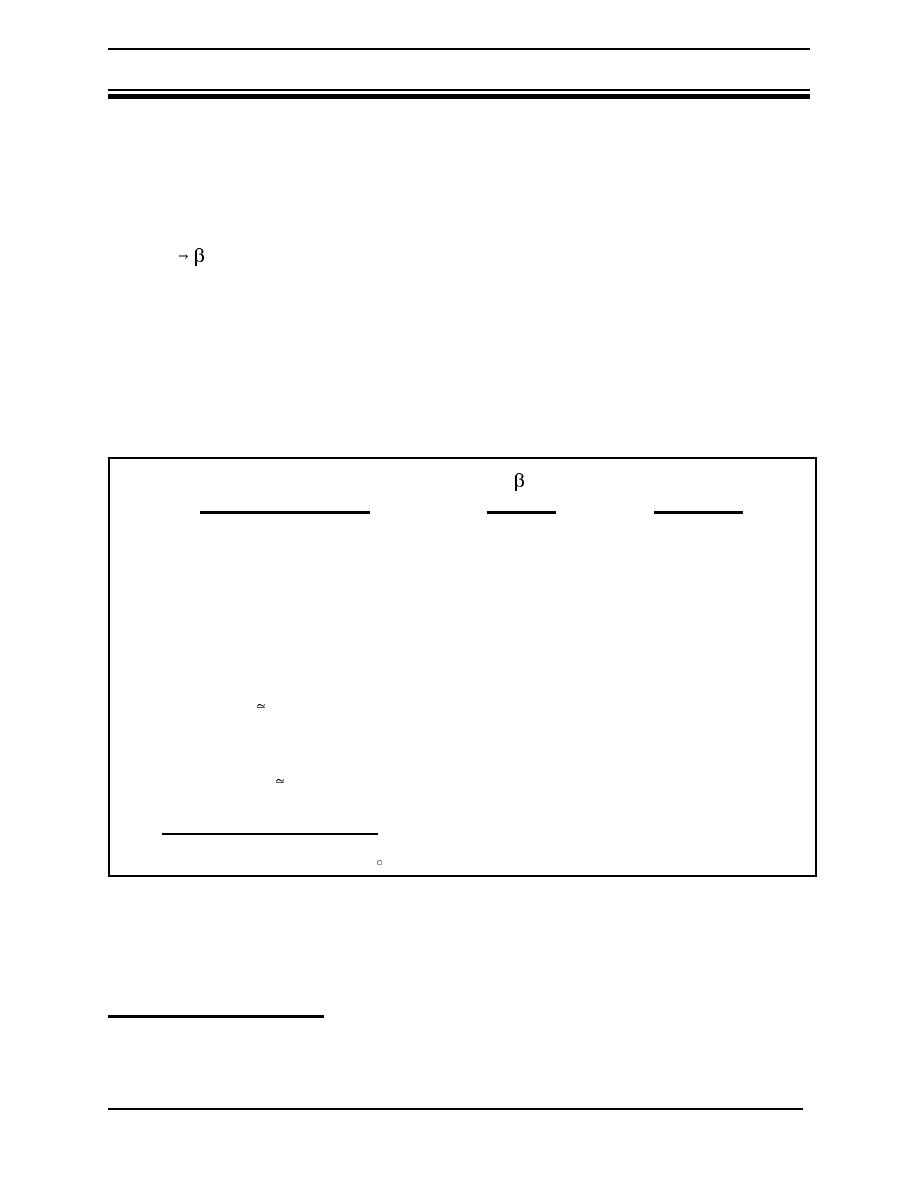
Table 3 Penetration depths of tritium betas |
|
||
| ||||||||||
|
|  PHYSICAL AND CHEMICAL PROPERTIES
DOE-HDBK-1079-94
Tritium Primer
beta particle may lose several electron volts (keV)b of energy, and the electron is stripped from its
atom (ionization) or promoted to an excited state. The beta particle has a finite penetration depth
that depends on its energy.
Recall that tritium undergoes beta decay according to the following equation:
3
+ 3He + energy .
H
1
2
The helium daughter ( 3He) is stable, but lighter than common helium ( 2He). The decay energy
4
2
is constant (18.6 keV), but is shared between the beta particle and an antineutrino (a tiny particle).
The result is that not all beta particles have the same energy. The average energy is 5.7 keV.
Consequently, not all tritium betas have the same penetration depth in a given material. Where
beta ranges are given, it is customary to list both the highest energy and the average, most
representative energy, as listed in Table 3.
Table 3 Penetration depths of tritium betas
E( )
Penetrati
Material
(keV)
on Depth
T2 gas, STPa
5.7
0.26 cm
T2 gas, STP
18.6
3.2 cm
Air, STP
5.7
0.036 cm
Air, STP
18.6
0.45 cm
Water, soft tissue
5.7
0.42 m
(and oils/polymers of
density 1)
Water, soft tissue
18.6
5.2 m
(and oils/polymers
of density 1)
Stainless steel
5.7
0.06 m
a. STP = Standard temperature (0 C) and pressure (760 Torr).
b. An electron volt is a small unit of energy used in descriptions of nuclear and chemical reactions.
It equals the energy gained by an electron when it moves across a potential of 1 volt.
Tritium
Page 12
Rev. 0
|
|
Privacy Statement - Press Release - Copyright Information. - Contact Us |