 |
|||
|
|
|||
|
Page Title:
Table C-1. Table of Tritium Compressibility Factors at 295 K (continued) |
|
||
| ||||||||||
|
| 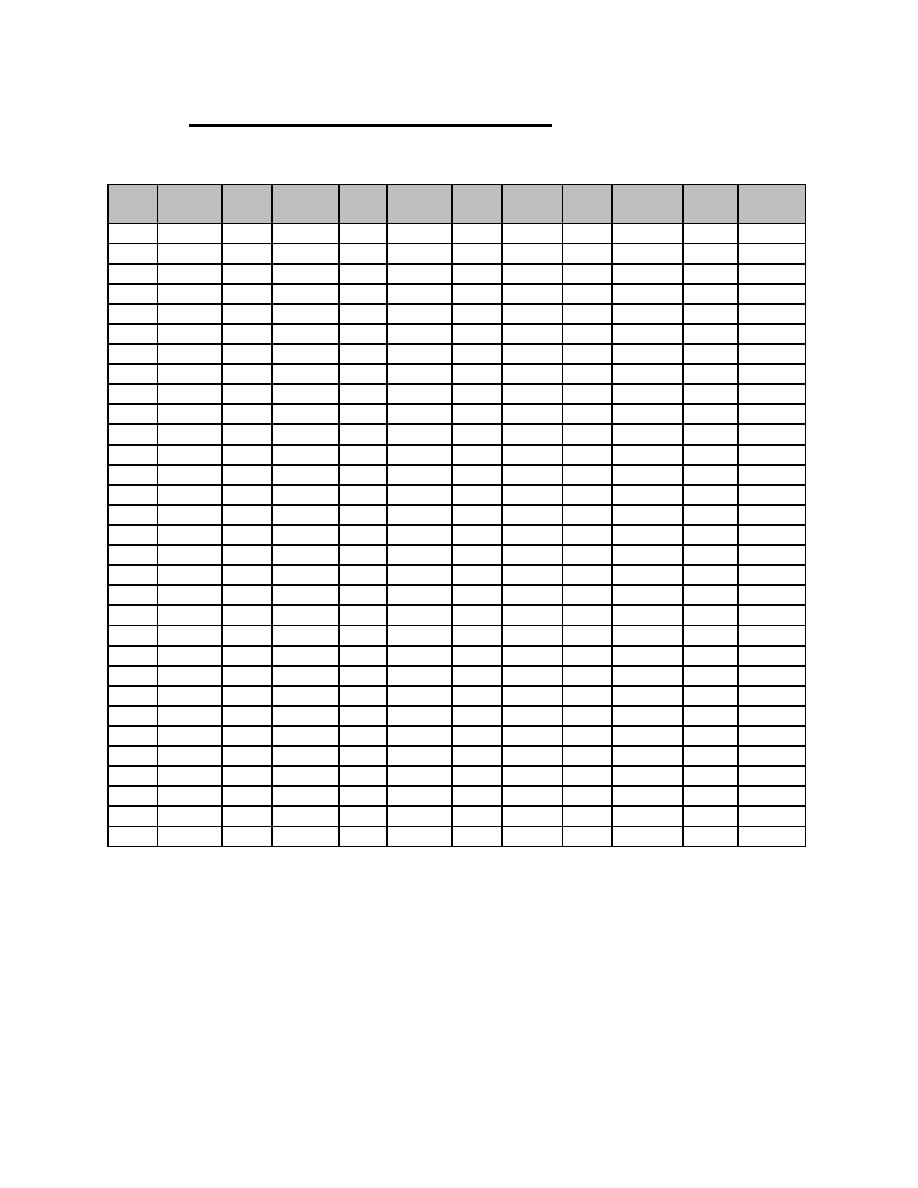 DOE-HDBK-1129-99
Table C-1. Table of Tritium Compressibility Factors at 295 K (continued)
Z(T2) = 1 + {[(Patm x 760) x 0.000832]/1000}
P
z(T2)
P
z(T2)
P
z(T2)
P
z(T2)
P
z(T2)
P
z(T2)
(atm)
(atm)
(atm)
(atm)
(atm)
(atm)
18.0
1.0114
21.0
1.0133
24.0
1.0152
27.0
1.0171
30.0
1.0190
33.0
1.0209
18.1
1.0114
21.1
1.0133
24.1
1.0152
27.1
1.0171
30.1
1.0190
33.1
1.0209
18.2
1.0115
21.2
1.0134
24.2
1.0153
27.2
1.0172
30.2
1.0191
33.2
1.0210
18.3
1.0116
21.3
1.0135
24.3
1.0154
27.3
1.0173
30.3
1.0192
33.3
1.0211
18.4
1.0116
21.4
1.0135
24.4
1.0154
27.4
1.0173
30.4
1.0192
33.4
1.0211
18.5
1.0117
21.5
1.0136
24.5
1.0155
27.5
1.0174
30.5
1.0193
33.5
1.0212
18.6
1.0118
21.6
1.0137
24.6
1.0156
27.6
1.0175
30.6
1.0193
33.6
1.0212
18.7
1.0118
21.7
1.0137
24.7
1.0156
27.7
1.0175
30.7
1.0194
33.7
1.0213
18.8
1.0119
21.8
1.0138
24.8
1.0157
27.8
1.0176
30.8
1.0195
33.8
1.0214
18.9
1.0120
21.9
1.0138
24.9
1.0157
27.9
1.0176
30.9
1.0195
33.9
1.0214
19.0
1.0120
22.0
1.0139
25.0
1.0158
28.0
1.0177
31.0
1.0196
34.0
1.0215
19.1
1.0121
22.1
1.0140
25.1
1.0159
28.1
1.0178
31.1
1.0197
34.1
1.0216
19.2
1.0121
22.2
1.0140
25.2
1.0159
28.2
1.0178
31.2
1.0197
34.2
1.0216
19.3
1.0122
22.3
1.0141
25.3
1.0160
28.3
1.0179
31.3
1.0198
34.3
1.0217
19.4
1.0123
22.4
1.0142
25.4
1.0161
28.4
1.0180
31.4
1.0199
34.4
1.0218
19.5
1.0123
22.5
1.0142
25.5
1.0161
28.5
1.0180
31.5
1.0199
34.5
1.0218
19.6
1.0124
22.6
1.0143
25.6
1.0162
28.6
1.0181
31.6
1.0200
34.6
1.0219
19.7
1.0125
22.7
1.0144
25.7
1.0163
28.7
1.0181
31.7
1.0200
34.7
1.0219
19.8
1.0125
22.8
1.0144
25.8
1.0163
28.8
1.0182
31.8
1.0201
34.8
1.0220
19.9
1.0126
22.9
1.0145
25.9
1.0164
28.9
1.0183
31.9
1.0202
34.9
1.0221
20.0
1.0126
23.0
1.0145
26.0
1.0164
29.0
1.0183
32.0
1.0202
35.0
1.0221
20.1
1.0127
23.1
1.0146
26.1
1.0165
29.1
1.0184
32.1
1.0203
35.1
1.0222
20.2
1.0128
23.2
1.0147
26.2
1.0166
29.2
1.0185
32.2
1.0204
35.2
1.0223
20.3
1.0128
23.3
1.0147
26.3
1.0166
29.3
1.0185
32.3
1.0204
35.3
1.0223
20.4
1.0129
23.4
1.0148
26.4
1.0167
29.4
1.0186
32.4
1.0205
35.4
1.0224
20.5
1.0130
23.5
1.0149
26.5
1.0168
29.5
1.0187
32.5
1.0206
35.5
1.0224
20.6
1.0130
23.6
1.0149
26.6
1.0168
29.6
1.0187
32.6
1.0206
35.6
1.0225
20.7
1.0131
23.7
1.0150
26.7
1.0169
29.7
1.0188
32.7
1.0207
35.7
1.0226
20.8
1.0132
23.8
1.0150
26.8
1.0169
29.8
1.0188
32.8
1.0207
35.8
1.0226
20.9
1.0132
23.9
1.0151
26.9
1.0170
29.9
1.0189
32.9
1.0208
35.9
1.0227
21.0
1.0133
24.0
1.0152
27.0
1.0171
30.0
1.0190
33.0
1.0209
36.0
1.0228
To determine the component in the gas and the number of moles of each component in the gas,
a sample of the container gas is analyzed, and the mole percent of each gas is determined.
This gas analysis results in a number for each component in the mixture, which represents the
mole percent of each gas at the time of the analysis. The mole percent (m%) is calculated by
m%(component) = (Moles of a component/Moles Total) x 100
Therefore, the Mole Percent Total (m%(Total)) is
m%(Total) = 100 = m%(T2)+ m%(HT)+ m%(DT)+ m%(CT4)+ m%(qTw) + m%(He-3)+ m%(N2)+ m%(O2) +
m% (etc.)
C-4
|
|
Privacy Statement - Press Release - Copyright Information. - Contact Us |